Introduction
Absorption/Reflectance spectroscopy in the UV-Visible region (200nm-800nm) is called UV-Vis spectroscopy.
This helps in understanding the observed colors of compounds. Basically, light is made to fall on the sample, and the intensity of the transmitted or reflected light is measured as a function of wavelength.
Experimental setup and components:
The instrument used in UV-Vis spectroscopy is called a UV-Vis spectrophotometer.
The following diagram shows the components of a typical spectrophotometer.
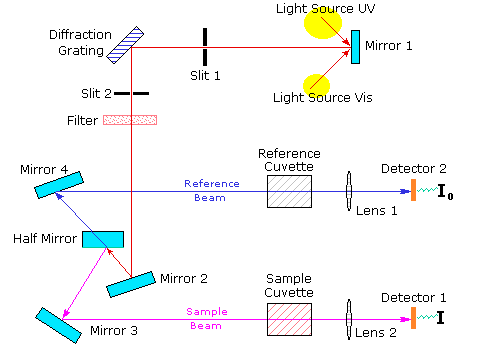
The functioning of this instrument is relatively straightforward. A beam of light from a visible and/or UV light source (colored red) is separated into its component wavelengths by a prism or diffraction grating. Each monochromatic (single wavelength) beam in turn is split into two equal intensity beams by a half-mirrored device. One beam, the sample beam (colored magenta), passes through a small transparent container (cuvette) containing a solution of the compound being studied in a transparent solvent. The other beam, the reference (colored blue), passes through an identical cuvette containing only the solvent. The intensities of these light beams are then measured by electronic detectors and compared. The intensity of the reference beam, which should have suffered little or no light absorption, is defined as . The intensity of the sample beam is defined as
. Over a short period of time, the spectrometer automatically scans all the component wavelengths in the manner described. The ultraviolet (UV) region scanned is normally from 200 to 400 nm, and the visible portion is from 400 to 800 nm
Light sources:
- Deuterium: D2 gas is discharged by contact with a high voltage tungsten cathode. Gives a continuous spectrum from ~150 nm -~370 nm. Usually used in conjunction with a tungsten/halogen source, which handles the visible spectrum.
- Xenon, Mercury/Xenon:
Flash Arc-Lamps –light generated from Xe plasma.
Pure Xenon has very wide emission spectrum ~200 –1200 nm. Xenon/Mercury is blue shifted for more power in the UV region.
Experimental measurements are usually done in terms of Transmittance , which is
where is the intensity of light that went through the sample and
is the intensity of the incident beam.
Observations can also be made in terms of Absorbance which is defined as:
Modern absorption instruments can usually display the data as either %-transmittance, or absorbance.
Principle (Theory)
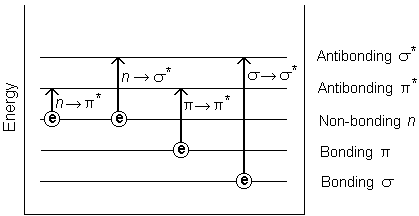
The absorption of UV or visible radiation corresponds to the excitation of outer electrons. When a photon is incident on the sample, then it’s energy can be absorbed to excite the electron to an excited state. Since the energy levels of photon and electrons are quantized, therefore only certain transitions are possible. There are three types of electronic transition which can be considered:
- Transitions involving
,
, and
electrons
- Transitions involving charge-transfer electrons
- Transitions involving d and f electrons
When an atom or molecule absorbs energy, electrons are promoted from their ground state to an excited state. In a molecule, the atoms can rotate and vibrate with respect to each other. These vibrations and rotations also have discrete energy levels, which can be considered as being packed on top of each electronic level.
Absorbing species containing  ,
,  , and
, and  electrons
electrons
Absorption of ultraviolet and visible radiation in organic molecules is restricted to certain functional groups (chromophores) that contain valence electrons of low excitation energy. The spectrum of a molecule containing these chromophores is complex. This is because the superposition of rotational and vibrational transitions on the electronic transitions gives a combination of overlapping lines. This appears as a continuous absorption band.
[Note: A chromophore is a chemical group which absorbs UV-visible radiation at a specific wavelength, with little influence from the other groups in the molecule. Typical chromophores in organic molecules are C=C double bonds, C=O carboxylic groups and aromatic rings. Only when two or more of these groups are conjugated, a relevant change in their absorption properties is observed.]
Possible electronic transitions of ,
, and
electrons are:
1. 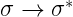 Transitions:
Transitions:
An electron in a bonding orbital is excited to the corresponding antibonding orbital. The energy required is large. For example, methane (which has only C-H bonds, and can only undergo
transitions) shows an absorbance maximum at 125 nm. Absorption maxima due to
transitions are not seen in typical UV-Vis. spectra (200 – 700 nm)
2.  Transitions:
Transitions:
Saturated compounds (compounds that have a chain of carbon atoms linked together by single bonds) containing atoms with lone pairs (non-bonding electrons) are capable of transitions. These transitions usually need less energy than
transitions. They can be initiated by light whose wavelength is in the range 150 – 250 nm. The number of organic functional groups with
peaks in the UV region is small.
3. 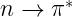 and
and  Transitions:
Transitions:
Most absorption spectroscopy of organic compounds is based on transitions of or
electrons to the
excited state. This is because the absorption peaks for these transitions fall in an experimentally convenient region of the spectrum (200 – 700 nm). These transitions need an unsaturated (containing double/triple bonds) group in the molecule to provide the
electrons.
Molar absorbtivities from transitions are relatively low, and range from 10 to 100 L mol-1 cm-1 .
transitions normally give molar absorbtivities between 1000 and 10,000 L mol-1 cm-1.
The solvent in which the absorbing species is dissolved also has an effect on the spectrum of the species. Peaks resulting from transitions are shifted to shorter wavelengths (blue shift) with increasing solvent polarity. This arises from increased solvation of the lone pair, which lowers the energy of the
orbital. Often (but not always), the reverse (i.e. red shift) is seen for
transitions. This is caused by attractive polarisation forces between the solvent and the absorber, which lower the energy levels of both the excited and unexcited states. This effect is greater for the excited state, and so the energy difference between the excited and unexcited states is slightly reduced – resulting in a small red shift. This effect also influences
transitions but is overshadowed by the blue shift resulting from solvation of lone pairs.
-> Charge – Transfer Absorption
Many inorganic species show charge-transfer absorption and are called charge-transfer complexes. For a complex to demonstrate charge-transfer behaviour, one of its components must have electron donating properties and another component must be able to accept electrons. Absorption of radiation then involves the transfer of an electron from the donor to an orbital associated with the acceptor.
Molar absorbtivities from charge-transfer absorption are large (greater that 10,000 L mol-1 cm-1).
Beer Lambert’s Law
Beer-Lambert’s law states that the Transmittance which is the ratio of the intensity of the transmitted light and the intensity of the incident light
, depends on the path length of light through the sample
, the absorption cross section
of the sample’s transition, and the difference in the population of the initial state
and final state
.
Another more popular form of Beer-Lambert’s law states that the Absorbance is directly proportional to the concentration of the absorbing species and the path length
.
wherer is the proportionality constant called the molar absorptivity.
The Beer-Lambert Law is useful for characterizing many compounds but does not hold as a universal relationship for the concentration and absorption of all substances. A 2nd order polynomial relationship between absorption and concentration is sometimes encountered for very large, complex molecules such as organic dyes (Xylenol Orange or Neutral Red, for example).
Derivation
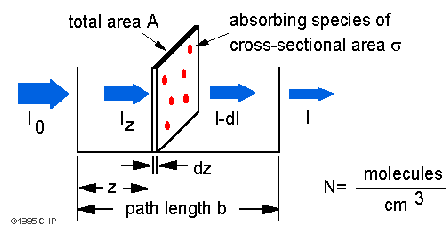
The Beer-Lambert law can be derived from an approximation for the absorption coefficient for a molecule by approximating the molecule to be an opaque disk of cross-sectional area which is the effective area seen by a photon of frequency
. Take a slab of infinitesimal width (dz) of sample.
is the intensity entering the sample at
,
is the intensity entering the infinitesimal slab at
,
is the intensity absorbed in the slab, and
is the intensity of light leaving the sample. Then, the total opaque area on the slab due to the absorbers is
. (Since
is the number of molecules per unit volume.) Then, the fraction of photons absorbed will be
so,
Integrating from z=0 to z=b
Converting number density to concentration(mol/litre) by multiplying by 1000cm^3 and dividing by Avogadro’s number,
Since N (molecules/cm3) * (1 mole / 6.023×1023 molecules) * 1000 cm3 / liter = c (moles/liter)
Using
where
Measuring semiconductor band gaps using UV-Vis Spectroscopy
For semiconductors, UV-vis spectroscopy offers a convenient method of estimating the optical band gap, since it probes electronic transitions between the valence band and the conduction band. The optical band gap is not necessarily equal to the electronic band gap, which is defined as the energy difference between the valence band minimum (VBM) and the conduction band maximum (CBM); however, it is often approximated as such because there are few convenient methods for measuring the electronic band gap. Exciton binding energies, d-d transitions, phonon absorption and emissions, and excitations to or from defect bands and color centers can complicate interpretation of UV-vis spectra; nevertheless, an estimation of the optical band gap is obtainable. Furthermore, UV-vis allows for the characterization of this electronic transition as either direct or indirect and whether it is allowed or forbidden.
A direct transition is described as a two-particle interaction between an electron and a photon, whereas an indirect transition is described as a three-particle interaction (photon, electron, phonon) to ensure momentum conservation. A transition is allowed or forbidden depending on the dipole selection rules associated with the system. The shape of the UV-vis absorption spectrum can distinguish between these transitions.
Applications
- Detection of functional groups- UV spectroscopy is used to detect the presence or absence of chromophore in the compound. This is technique is not useful for the detection of chromophore in complex compounds. The absence of a band at a particular band can be seen as an evidence for the absence of a particular group. If the spectrum of a compound comes out to be transparent above 200 nm than it confirms the absence of –
a) Conjugation b) A carbonyl group c) Benzene or aromatic compound d) Bromo or iodo atoms. -
Detection of extent of conjugation- The extent of conjugation in the polyenes can be detected with the help of UV spectroscopy. With the increase in double bonds the absorption shifts towards the longer wavelength. If the double bond is increased by 8 in the polyenes then that polyene appears visible to the human eye as the absorption comes in the visible region.
-
Identification of an unknown compound- An unknown compound can be identified with the help of UV spectroscopy. The spectrum of unknown compound is compared with the spectrum of a reference compound and if both the spectrums coincide then it confirms the identification of the unknown substance.
-
Determination of configurations of geometrical isomers- It is observed that cis-alkenes absorb at different wavelength than the trans-alkenes. The two isomers can be distinguished with each other when one of the isomers has non-coplanar structure due to steric hindrances. The cis-isomer suffers distortion and absorbs at lower wavelength as compared to trans-isomer.
-
Determination of the purity of a substance- Purity of a substance can also be determined with the help of UV spectroscopy. The absorption of the sample solution is compared with the absorption of the reference solution. The intensity of the absorption can be used for the relative calculation of the purity of the sample substance.
-
Measuring the thickness of films
-
Measuring the direct/indirect band-gap of semiconductors
Limitations of UV-Vis Spectroscopy
- Deviation from Beer-Lambert’s law
The linearity of the Beer-Lambert law is limited by chemical and instrumental factors. Causes of nonlinearity include:
-> deviations in absorptivity coefficients at high concentrations (>0.01M) due to electrostatic interactions between molecules in close proximity
-> scattering of light due to particulates in the sample
-> fluoresecence or phosphorescence of the sample
-> changes in refractive index at high analyte concentration
-> shifts in chemical equilibria as a function of concentration
->non-monochromatic radiation, deviations can be minimized by using a relatively flat part of the absorption spectrum such as the maximum of an absorption band
-> stray light
- Measuring the band-gap
To derive a band gap value from a UV-vis measurement, the data must be interpreted. Interpretation is often made difficult by the shape of the absorption spectrum and the ability of the user to estimate the line tangent to
the slope of the absorption data. This procedure requires the drawing of a tangent line to the curve, which is subjective and can result in significant error.
- Stray light
Other sources of error in a UV-vis measurement often arise from reflection,
refraction, or scattering that may occur at the surface and interfaces of the material or
from the unwanted transmission of light during a diffuse reflectance experiment. These
effects decrease the amount of light that reaches the detector and produce seemingly
higher absorption values. This can result in nonzero baselines or sloped base lines that
need to be taken into account when analyzing spectra. To minimize reflection or
refraction during a transmission experiment, the user should ensure that the sample sits
normal to the path of incident light. Scattering effects can be minimized by placing the
sample as close as possible to the detector. In a diffuse reflectance measurement, the user
should place a highly reflective standard against a transparent sample to decrease
transmitted light.
Pitfalls of the experimental procedure often come from an improperly positioned
sample, as mentioned above, or from measurements performed before the lamp has had
proper time to warm up, resulting in an unreliable base line. Improper shielding of the
sampling chamber from ambient lighting can also contribute to the background signal and
decrease the signal-to-noise ratio. Lastly, harmonics that arise from using a grating
monochromator can lead to inaccurate measurements if they are not removed using longpass-
filters.
References:
https://en.wikipedia.org/wiki/Ultraviolet–visible_spectroscopy
https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/spectrpy/uv-vis/spectrum.htm
https://www2.chemistry.msu.edu/faculty/reusch/virttxtjml/spectrpy/uv-vis/uvspec.htm
http://life.nthu.edu.tw/~labcjw/BioPhyChem/Spectroscopy/beerslaw.htm
https://teaching.shu.ac.uk/hwb/chemistry/tutorials/molspec/uvvisab1.htm
https://en.wikipedia.org/wiki/Beer–Lambert_law
http://www.chemistry.iitkgp.ac.in/faculty/SDG/Spectroscopy%20I.pdf
I’m a physicist specializing in computational material science with a PhD in Physics from Friedrich-Schiller University Jena, Germany. I write efficient codes for simulating light-matter interactions at atomic scales. I like to develop Physics, DFT, and Machine Learning related apps and software from time to time. Can code in most of the popular languages. I like to share my knowledge in Physics and applications using this Blog and a YouTube channel.
